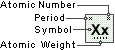Antimony
Symbol
Sb
Atomic Number
51
Atomic Weight
121.760
Element Classification
Discovered By
Known to the ancients.
Discovery Date
n/a (Unknown)
Name Origin
Greek: anti and monos (not alone), symbol from mineral stibnite.
Density (g/cc)
6.691
Melting Point (K)
903.9
Boiling Point (K)
1908
Appearance
Hard, silvery-white, brittle semimetal
Atomic Radius (pm)
159
Atomic Volume (cc/mol)
18.4
Covalent Radius (pm)
140
Ionic Radius
62 (+6e) 245 (-3)
Specific Heat
0.205
Fusion Heat (kJ/mol)
20.08
Evaporation Heat (kJ/mol)
195.2
Thermal Conductivity (@25°C W/m K)
24.43
Debye Temperature (K)
200.00
Pauling Negativity Number
2.05
First Ionizing Energy (kJ/mol)
833.3
Oxidation States
5, 3, -2
Electronic Configuration
[Kr] 4d10 5s2 5p3
Lattice Structure
Rhombohedral(RHL)
Lattice Constant
4.510
Lattice C/A Ratio
n/a