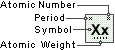Zirconium
Symbol
Zr
Atomic Number
40
Atomic Weight
91.224
Element Classification
Discovered By
Martin Klaproth
Discovery Date
1789 (Germany)
Name Origin
The mineral, zircon.
Density (g/cc)
6.506
Melting Point (K)
2125
Boiling Point (K)
4650
Appearance
Gray-white, lustrous, corrosion-resistant metal
Atomic Radius (pm)
160
Atomic Volume (cc/mol)
14.1
Covalent Radius (pm)
145
Ionic Radius
79 (+4e)
Specific Heat
0.281
Fusion Heat (kJ/mol)
19.2
Evaporation Heat (kJ/mol)
567
Thermal Conductivity (@25°C W/m K)
22.7
Debye Temperature (K)
250.00
Pauling Negativity Number
1.33
First Ionizing Energy (kJ/mol)
659.7
Oxidation States
4
Electronic Configuration
[Kr] 4d2 5s2
Lattice Structure
Hexagonal(HEX)
Lattice Constant
3.230
Lattice C/A Ratio
1.593