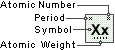Praseodymium
Symbol
Pr
Atomic Number
59
Atomic Weight
140.90765
Element Classification
Discovered By
C.F. Aver von Welsbach
Discovery Date
1885 (Austria)
Name Origin
Greek: prasios and didymos (green twin), from its green salts.
Density (g/cc)
6.773
Melting Point (K)
1204
Boiling Point (K)
3785
Appearance
Silvery white, moderately soft, malleable, and ductile metal
Atomic Radius (pm)
182
Atomic Volume (cc/mol)
20.8
Covalent Radius (pm)
165
Ionic Radius
90 (+4e) 101.3 (+3e)
Specific Heat
0.192
Fusion Heat (kJ/mol)
11.3
Evaporation Heat (kJ/mol)
331
Thermal Conductivity (@25°C W/m K)
12.5
Debye Temperature (K)
n/a
Pauling Negativity Number
1.13
First Ionizing Energy (kJ/mol)
526.6
Oxidation States
4, 3
Electronic Configuration
[Xe] 4f3 6s2
Lattice Structure
Hexagonal(HEX)
Lattice Constant
3.670
Lattice C/A Ratio
1.614