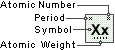Strontium
Symbol
Sr
Atomic Number
38
Atomic Weight
87.62
Element Classification
Discovered By
A. Crawford
Discovery Date
1790 (Scotland)
Name Origin
The Scottish town, Strontian.
Density (g/cc)
2.54
Melting Point (K)
1042
Boiling Point (K)
1657
Appearance
Silvery, malleable metal
Atomic Radius (pm)
215
Atomic Volume (cc/mol)
33.7
Covalent Radius (pm)
191
Ionic Radius
112 (+2e)
Specific Heat
0.301
Fusion Heat (kJ/mol)
9.20
Evaporation Heat (kJ/mol)
144
Thermal Conductivity (@25°C W/m K)
(35.4)
Debye Temperature (K)
n/a
Pauling Negativity Number
0.95
First Ionizing Energy (kJ/mol)
549.0
Oxidation States
2
Electronic Configuration
[Kr] 5s2
Lattice Structure
Face-Centered Cubic(FCC)
Lattice Constant
6.080
Lattice C/A Ratio
n/a